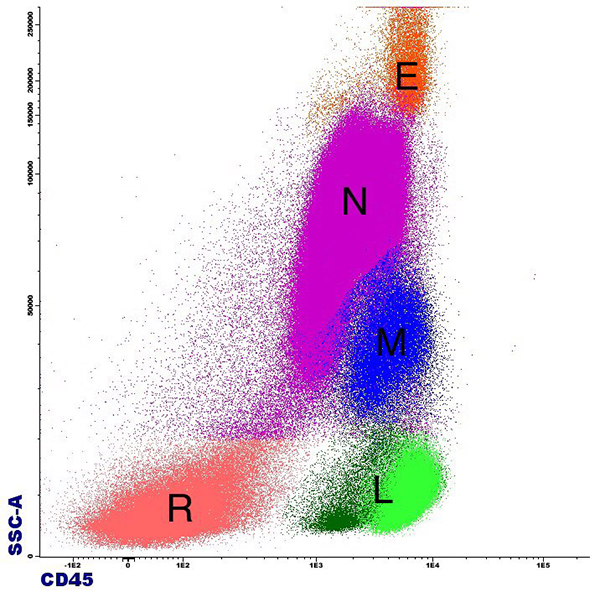
Multicolour Flowcytometry or Immunophenotyping
Bone Marrow Reporting | Bone Marrow Biopsy
Flow cytometry is an highly advance technique of cell analysis commonly used in laboratory hematology and immunology. Flow cytometry is used for immunophenotyping of a variety of specimens, including whole blood, bone marrow, serous cavity fluids, cerebrospinal fluid, urine, and solid tissues.
MRD(Minimal Residual Disease) diagnostics by Flowcytometry has proven to be the strongest prognostic factor, allowing for risk group assignment into different treatment arms, ranging from low-risk/standard-risk Leukaemia with treatment reduction to medium-risk or high-risk with mild or strong intensification, respectively.
Services Available:
-
Diagnosis of Acute leukaemia [B-ALL, T-ALL, AML]
-
Diagnosis of Lymphoproliferative disorder or Lymphoma (CLL, Hairy cell Leukaemia, T-LGL & others
-
Minimal Residual Disease ( MRD ) for Leukaemia by Flowcytometry (B- ALL, T- ALL, AML, CLL, HCL)
-
Diagnosis of PNH (Paroxysmal Nocturnal Hemoglobinuria) – FLAER, CD55,CD59
-
CD4 & CD8 count for HIV monitoring
-
HLA B 27 for Ankylosing spondylitis
-
CD34 by ISHAGE for Stem cell or Bone marrow Transplant
-
Lymphocyte subset analysis for Immunodeficiency disorders.
-
EMA dye binding test for Hereditary spherocytosis
-
Platelet function tests by flowcytometety
-
CD3,CD20,CD19,CD22 count for Drug (like Rituximab)
HaemPath Diagnostics Centre, NABL Accrediated & ISO Certified Gujarat's 1st Super Speciality Lab, provide 13 colour flowcytometery or Immunophenotyping diagnostic services.
Quick Contact
How Can We Help?
If you need any helps, please feel free to contact us.
Multicolor Flowcytometry | Minimal Residual Disease
| Test Code | Test Name | Method | Sample type | Anticoagulant | Voume | MRP | Ambient stability | Shipping Temp | Turnaround time |
|---|---|---|---|---|---|---|---|---|---|
| HP001 | Immunophenotyping for Leukaemia/CLPD | Flowcytometry | Blood/ Bone marrow / fluids | EDTA/HEPARIN | 3ML | 9000 | 1 D | A | 24 hrs |
| HP002 | Minimal Residual Disease ( MRD ) – B ALL | Flowcytometry | Bone marrow | EDTA/HEPARIN | 3 ML | 10000 | 1 D | A | 24 hrs |
| HP003 | Minimal Residual Disease ( MRD )– T ALL | Flowcytometry | Bone marrow | EDTA/HEPARIN | 3 ML | 10000 | 1 D | A | 24 hrs |
| HP004 | Minimal Residual Disease ( MRD ) – AML | Flowcytometry | Bone marrow | EDTA/HEPARIN | 3 ML | 12500 | 1 D | A | 24 hrs |
| HP005 | Minimal Residual Disease ( MRD ) – Myeloma | Flowcytometry | Bone marrow | EDTA/HEPARIN | 3 ML | 10000 | 1 D | A | 24 hrs |
| HP006 | Minimal Residual Disease ( MRD ) – CLL | Flowcytometry | Bone marrow | EDTA/HEPARIN | 3 ML | 10000 | 1 D | A | 24 hrs |
| HP007 | Minimal Residual Disease ( MRD ) – Hairy Cell Leukaemia | Flowcytometry | Bone marrow | EDTA/HEPARIN | 3 ML | 10000 | 1 D | A | 24 hrs |
| HP008 | PNH (CD55,CD59,FLAER,CD157,CD33,CD45) | Flowcytometry | Peripheral blood | EDTA/HEPARIN | 3 ML | 6000 | 1 D | A | 8 hrs |
| HP009 | PNH for FLAER ( FLAER,CD157,CD33,CD45) | Flowcytometry | Peripheral blood | EDTA/HEPARIN | 3 ML | 4000 | 1 D | A | 8 hrs |
| HP010 | Lymphocyte subset analysis | Flowcytometry | Peripheral blood | EDTA/HEPARIN | 3 ML | 4500 | 1 D | A | 24 hrs |
| HP011 | CD34 count by ISHAGE protocol | Flowcytometry | Peripheral blood/Harvest Product | EDTA/HEPARIN | 3 ML | 4500 | 1 D | A | 6 hrs. |
| HP012 | CD4 & CD8 count | Flowcytometry | Peripheral blood | EDTA/HEPARIN | 3 ML | 2000 | 1 D | A | 8 hrs |
| HP013 | NK cell estimation | Flowcytometry | Peripheral blood | EDTA/HEPARIN | 3 ML | 3200 | 1 D | A | 8 hrs |
| HP014 | CD19 count | Flowcytometry | Peripheral blood | EDTA/HEPARIN | 3 ML | 2000 | 1 D | A | 8 hrs |
| HP015 | CD 20 count | Flowcytometry | Peripheral blood | EDTA/HEPARIN | 3 ML | 2000 | 1 D | A | 8 hrs |
| HP016 | CD 3 count | Flowcytometry | Peripheral blood/Harvest product | EDTA/HEPARIN | 3 ML | 4000 | 1 D | A | 8 hrs |
| HP017 | HLA B27 by flowcytometry | Flowcytometry | Peripheral blood | EDTA/HEPARIN | 3 ML | 1800 | 1 D | A | 8 hrs |
| HP018 | PLATELET DISORDERS (GT,BSS) | Flowcytometry | Peripheral blood | CITRATE | 5 ML | 6500 | 1 D | A | 24 hrs |
| HP019 | EMA dye binding test for Hereditary spherocytosis | Flowcytometry | Peripheral blood | EDTA | 5 ML | 4000 | 1 D | A | 24 hrs |
| HP020 | LAD-1 for leukocyte adhesion deficiency | Flowcytometry | Peripheral blood | EDTA | 4 ML | 4000 | 1 D | A | 24 hrs |
Disclaimer
- The Directory of Services is effective from 1St July 2020.
- Rates are inclusive of all taxes. However, if government imposes service Tax, it will be charged additionally.
- Rate are subject to change without any prior notice/information.
- Information provided regarding investigations are given to best of our knowledge.
- In case of collected specimen, it is presumed that patient’s demographics are confirmed at the point of generation of the said specimen
- Under any unanticipated circumstances (non-availability of kits, instrument breakdown, natural calamities etc.), tests may not be reported as per schedule. Haempath Diagnostics will ensure that the delay is minimized.
- Though effort will be made to convey periodic updates, Haempath Diagnostics is not liable for not communicating changes or discontinuation of any tests.
A fresh specimen will be requested if testing cannot be performed for the following reason.
- Insufficient quantity of the specimen
- Specimen quality is unacceptable. [ Hemolysed, clotted ,lipemic etc.)
- Incorrect specimen type
- Incorrect identity of the specimen
Storage & Transportation
- Hemostasis & Thrombosis assays are very sensitive for preanalytical variables, storage & transportation.
- Outstation sample for Hemostasis & Thrombosis required transportation in Dry ice only.
- Flowcytometry sample should be stored & transported at room temperature (10 – 24 C).
- MRD assay required fresh & first pool sample.
- Outside laboratory collection for platelet function tests not acceptable except in special condition.
Note:
- Discussion with Laboratory personals required before sending special tests of specifically Coagulation & Thrombotic disorders.
- All special tests will be done on working day at Haempath Diagnostics except Sunday & holidays with prior appointment.





![Multicolor Flowcytometer [13 color]](images/single-img-ten2.jpg?v=4)